
Here’s a statistic that surprises most couples: in approximately 50% of infertility cases, a male factor is either the primary cause or a significant contributor. Yet in many clinical settings — particularly in South Asia, the Middle East, and parts of Europe — the default assumption is still that infertility is “a woman’s problem.” The woman undergoes months of hormonal investigations, ultrasound scans, and sometimes invasive procedures before anyone thinks to check the man’s semen analysis.
I see the consequences of this delay regularly. A couple arrives after two or three years of failed conception, carrying a folder full of the wife’s investigations. The husband has never had a single test. When I finally analyze his semen, the answer has been there all along — a clinical varicocele suppressing his sperm count, or an undiagnosed hormonal imbalance, or — in one memorable case — complete azoospermia (zero sperm) caused by testosterone injections prescribed by an online clinic without a single word about fertility consequences.
Male infertility is not a sentence. Many causes are treatable or correctable — if they’re actually identified. This article is the conversation I wish every man received before his partner was subjected to years of unnecessary investigation alone.
📋 Key Takeaways
- Male factors contribute to approximately 50% of all infertility cases — yet men are often the last to be investigated
- A semen analysis is the single most important first-line test — it should be performed early in any infertility work-up, not as an afterthought
- Varicocele (dilated testicular veins) is the most common correctable cause of male infertility, present in up to 40% of infertile men, and surgical repair improves semen parameters in 60–70% of cases
- Testosterone replacement therapy (TRT) shuts down sperm production — this is one of the most common iatrogenic causes of male infertility I see in clinic
- Sperm quality declines with age, but male fertility does not have a sharp “cliff” like female menopause — men can father children into their 60s and beyond, albeit with reduced efficiency and increased genetic risk
- Lifestyle factors — heat exposure, smoking, obesity, alcohol, and anabolic steroids — are all evidence-based, modifiable causes of reduced sperm quality
In This Guide:

The Semen Analysis: Your Most Important First Test
A semen analysis is cheap, non-invasive, and provides an enormous amount of clinical information. It should be one of the first investigations performed when a couple presents with infertility — ideally within the first month of the work-up. The fact that it’s often delayed for months or years reflects cultural stigma, not clinical logic.
The World Health Organization (WHO) 6th Edition (2021) reference values for semen analysis are:
Volume: ≥1.4 mL. Low volume may suggest ejaculatory duct obstruction, retrograde ejaculation, or incomplete collection.
Sperm concentration: ≥16 million per mL. Below this is termed oligozoospermia (low sperm count).
Total sperm count: ≥39 million per ejaculate.
Progressive motility: ≥30%. This measures the percentage of sperm swimming forward effectively. Below this is asthenozoospermia (poor motility).
Total motility: ≥42%.
Normal morphology: ≥4% (strict Kruger criteria). Below this is teratozoospermia (abnormal shape). This value shocks many men — 96% of sperm being “abnormal” is actually normal. The bar for what counts as morphologically perfect is extremely high.
Critical point: A single abnormal semen analysis does not constitute a diagnosis. Semen parameters fluctuate significantly — illness, fever, stress, medications, and even the time since last ejaculation can all affect results. The WHO and EAU guidelines require at least two analyses, 1–3 months apart, before drawing conclusions.
How to Produce an Accurate Sample
This matters more than most men realize. An improperly collected sample produces misleading results — typically falsely low. Follow these rules:
Abstinence window: 2–7 days without ejaculation before the test. Less than 2 days can give a low count; more than 7 days can reduce motility.
Complete collection: The first portion of the ejaculate contains the highest sperm concentration. If any is spilled, the results will be artificially low. Inform the lab if the collection was incomplete.
Container: Use a sterile, wide-mouthed container provided by the lab. Never use a condom — most contain spermicidal lubricant. If collection must occur at home, deliver the sample to the lab within 60 minutes, kept at body temperature (inside a jacket pocket, not in a bag exposed to cold).
Timing: Produce the sample preferably at the lab itself, if facilities allow. This eliminates transport-related degradation.
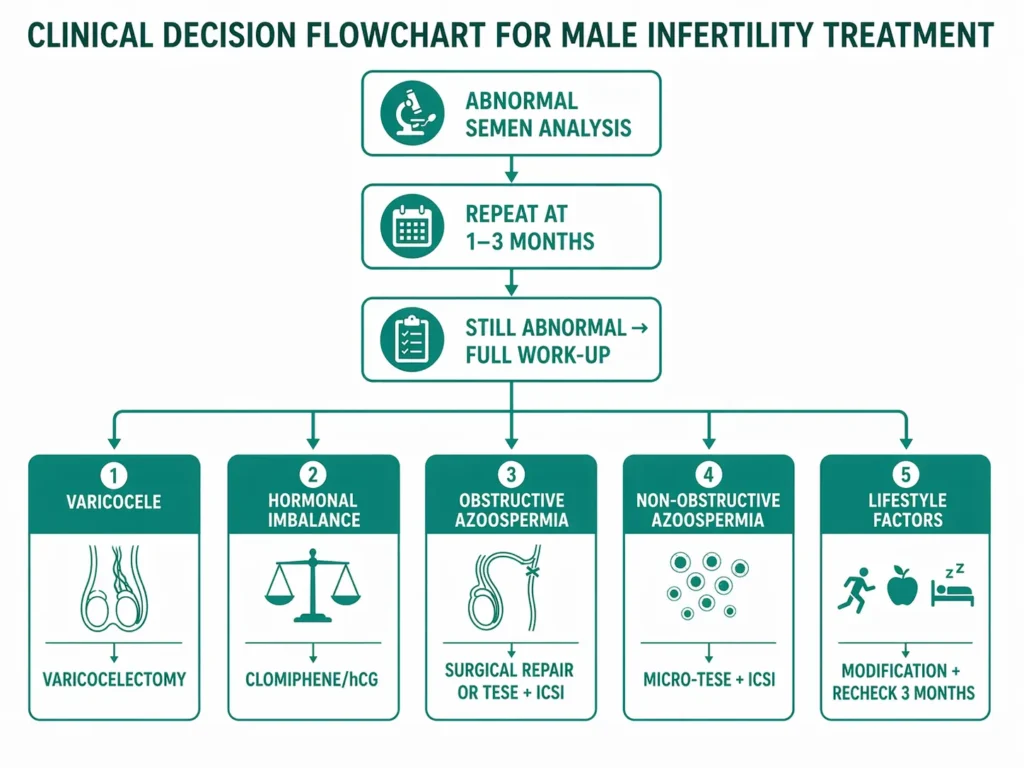
The Major Causes of Male Infertility
1. Varicocele — The Most Common Correctable Cause
A varicocele is an abnormal dilatation of the pampiniform venous plexus — the network of veins draining the testicle. Think of it as varicose veins of the scrotum. Varicoceles are present in approximately 15% of all men and up to 40% of men presenting with infertility. They’re far more common on the left side (85%) due to the anatomy of the left testicular vein, which drains vertically into the left renal vein (making it more susceptible to reflux).
The mechanism of damage is primarily heat. The pooled venous blood raises intrascrotal temperature by 1–2°C — enough to impair spermatogenesis (sperm production), which is temperature-sensitive and requires the scrotal environment to be 2–3°C below core body temperature. Varicoceles also increase oxidative stress and reduce testosterone production in the affected testis.
Surgical repair (varicocelectomy) improves semen parameters in 60–70% of men and leads to spontaneous pregnancy in approximately 30–40% of couples. The microsurgical subinguinal approach has the lowest recurrence rate (1–2%) and complication rate. I consider varicocelectomy for men with a clinically palpable varicocele, abnormal semen parameters, and a partner with normal or correctable female fertility.
2. Hormonal Imbalances
The hormonal cascade driving sperm production starts in the brain. The hypothalamus releases GnRH (gonadotropin-releasing hormone), which signals the pituitary to release LH (luteinizing hormone) and FSH (follicle-stimulating hormone). LH drives testosterone production in the Leydig cells. FSH, together with intratesticular testosterone, drives spermatogenesis in the Sertoli cells. A disruption at any level can impair fertility.
Hypogonadotropic hypogonadism (low LH and FSH) — caused by pituitary tumors, chronic opioid use, obesity, or exogenous testosterone — is potentially reversible. Remove the cause and spermatogenesis can recover. Hypergonadotropic hypogonadism (high LH and FSH with low testosterone) — seen in Klinefelter syndrome, testicular failure, or post-chemotherapy — is more difficult to treat as it reflects primary testicular damage.
➡️ Related: Low Testosterone in Men Over 40: Signs Your Doctor Might Miss
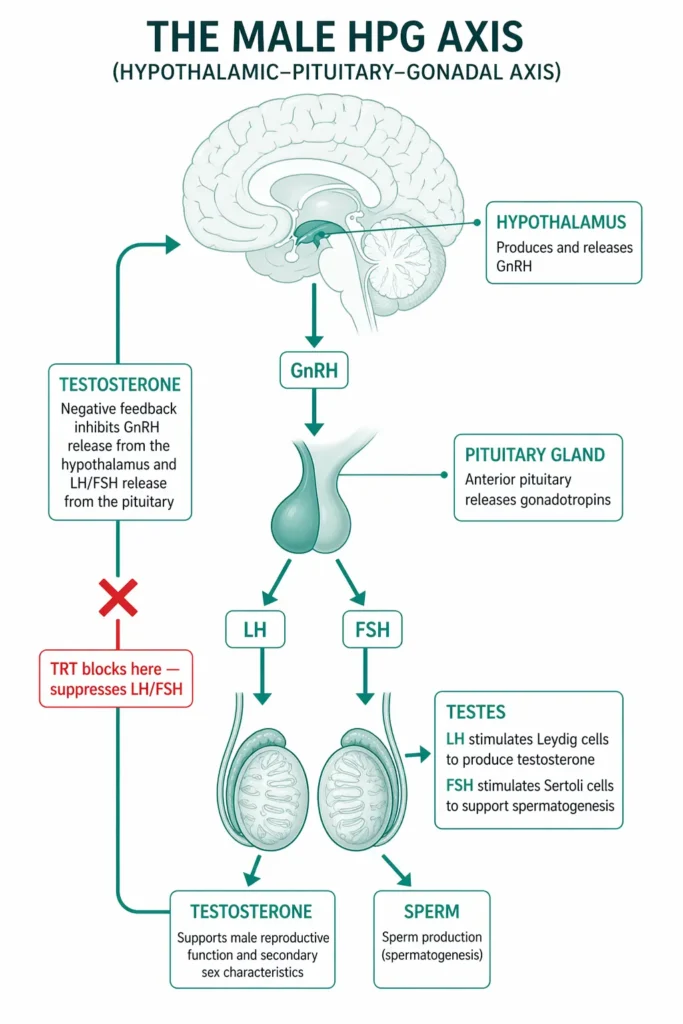
3. TRT-Induced Infertility — A Growing Iatrogenic Problem
This deserves its own section because it’s one of the most preventable causes of male infertility I encounter. When a man takes exogenous testosterone — whether prescribed by a doctor, obtained from an online TRT clinic, or self-administered from gym suppliers — his pituitary stops producing LH and FSH. Without FSH, Sertoli cells cannot support spermatogenesis. Within 3–6 months, most men become azoospermic.
The tragedy is that many of these men were not counseled about this effect. Some were prescribed TRT for “low energy” or “optimization” with testosterone levels in the normal range. Some were body builders using supraphysiological doses. In both cases, the pathway to recovery is the same: stop the testosterone, wait for the HPG axis to recover (6–12 months, sometimes longer), and consider hCG or clomiphene citrate to stimulate endogenous production during the recovery period.
Recovery is not guaranteed. A minority of men — particularly those who’ve used high doses for extended periods — develop persistent impairment of spermatogenesis even after TRT cessation. This is why the EAU guidelines explicitly state: TRT is contraindicated in men wishing to preserve fertility.
4. Obstructive Causes
Obstructive azoospermia — zero sperm in the ejaculate due to a physical blockage — accounts for approximately 40% of azoospermia cases. The testes are producing sperm normally, but the sperm cannot reach the ejaculate. Causes include: prior vasectomy, congenital bilateral absence of the vas deferens (CBAVD, associated with the CFTR gene mutations that cause cystic fibrosis), ejaculatory duct obstruction, and post-infectious scarring (e.g., from epididymitis or sexually transmitted infections).
The good news: obstructive azoospermia is often surgically correctable. Vasovasostomy (vasectomy reversal), vasoepididymostomy, and transurethral resection of the ejaculatory ducts (TURED) can restore sperm to the ejaculate. When surgical reconstruction isn’t possible, sperm can be retrieved directly from the testis or epididymis (TESE, micro-TESE, or PESA) for use with intracytoplasmic sperm injection (ICSI) in IVF.
5. Non-Obstructive Azoospermia — The Most Challenging Diagnosis
Non-obstructive azoospermia (NOA) means the testes are not producing sperm — or producing them in such small quantities that none reach the ejaculate. Causes include genetic conditions (Klinefelter syndrome, Y-chromosome microdeletions), cryptorchidism (undescended testes), post-chemotherapy or radiotherapy testicular failure, and idiopathic (unknown cause).
Even in NOA, there is hope. Micro-TESE (microsurgical testicular sperm extraction) — a procedure where the urologist uses an operating microscope to identify and extract individual foci of spermatogenesis within the testis — has a sperm retrieval rate of approximately 40–60% in experienced centers. These sperm can then be used with ICSI. For men with Klinefelter syndrome specifically, micro-TESE retrieval rates of 40–70% have been reported.
Lifestyle Factors That Damage Sperm — and What the Evidence Shows
Unlike the female oocyte (egg), which is fixed at birth, sperm are produced continuously — the full cycle of spermatogenesis takes approximately 74 days. This means that lifestyle exposures affecting sperm quality are potentially reversible within 3 months of eliminating the exposure. This is both an opportunity and a warning.

Heat Exposure
The testes are external to the body specifically because spermatogenesis requires a temperature 2–3°C below core body temperature. Anything that raises scrotal temperature impairs sperm production: tight underwear, prolonged laptop use on the lap, frequent hot baths or saunas, extended sitting (truck drivers, office workers), and heated car seats. A Finnish study showed that regular sauna use (>2 sessions/week) was associated with reduced sperm concentration and motility, but parameters recovered within 3 months of stopping.
Smoking
Cigarette smoking impairs every measurable semen parameter: concentration drops by 15–23%, motility by 10–17%, and morphology is worse in smokers versus non-smokers. The mechanism involves increased oxidative stress, heavy metal exposure (cadmium, lead), and direct DNA fragmentation of sperm. Cessation improves parameters over 3–6 months.
Obesity
Obesity impairs fertility through multiple mechanisms: increased aromatization of testosterone to estradiol (reducing intratesticular testosterone), elevated scrotal temperature from thigh fat, increased oxidative stress, and insulin resistance affecting Sertoli cell function. Men with a BMI above 30 have significantly lower sperm concentration and total motile sperm count compared to men with normal BMI.
Alcohol
Heavy alcohol consumption is directly toxic to Leydig cells and Sertoli cells, reducing both testosterone and sperm production. Moderate consumption appears to have minimal impact on fertility in most studies, though the data is not entirely consistent.
Anabolic Steroids
Anabolic androgenic steroids (AAS) — used by bodybuilders and increasingly by image-conscious younger men — suppress the HPG axis even more aggressively than therapeutic TRT. Supraphysiological doses cause azoospermia in over 90% of users within 3–6 months. Recovery after cessation is variable: most men recover spermatogenesis within 12 months, but a significant minority (up to 20%) have persistent impairment even years later, particularly after prolonged high-dose use.
➡️ Related: Is It ED or Heart Disease? Why Morning Wood Is a ‘Check Engine’ Light
Does Male Fertility Decline With Age?
Yes — but not in the binary way that female fertility does. Women have a finite reserve of oocytes that depletes to menopause. Men continue to produce sperm throughout life. However, sperm quality does decline with age, and this has measurable consequences:
Sperm volume and motility decrease gradually after age 40. Total motile sperm count declines by approximately 3% per year after age 40.
DNA fragmentation increases with age. Sperm from older men have higher rates of DNA damage, which is associated with reduced fertilization rates, lower implantation rates, higher miscarriage rates, and increased risk of de novo genetic mutations in offspring.
Time to conception increases. A large European study found that men over 40 took significantly longer to achieve pregnancy with a partner, even after adjusting for female age.
The practical implication: don’t assume unlimited time. While men don’t have a menopause, delaying fatherhood into the late 40s and 50s does carry biological consequences — both for conception rates and for the genetic health of offspring.
The Work-Up: What a Proper Male Fertility Evaluation Looks Like
A comprehensive male fertility work-up should include:
1. History: Duration of infertility, previous pregnancies (with current or previous partners), sexual history (frequency, timing, erectile/ejaculatory function), medical history (cryptorchidism, mumps, STIs, surgeries), medication history (TRT, anabolic steroids, opioids, finasteride, SSRIs), and lifestyle (smoking, alcohol, heat exposure, occupation).
2. Physical examination: Testicular size (normal: 15–25 mL by orchidometer), consistency, varicocele assessment (with and without Valsalva maneuver), epididymal fullness, vas deferens palpation (absent in CBAVD), and secondary sexual characteristics (gynecomastia, body hair distribution).
3. Semen analysis (x2): As described above — two analyses, 1–3 months apart, with correct abstinence and collection protocol.
4. Hormonal profile: Total testosterone (fasting, morning), FSH, LH, prolactin, estradiol, SHBG. FSH is particularly important: elevated FSH (>12 IU/L) suggests impaired spermatogenesis and correlates with reduced testicular reserve.
5. Ultrasound: Scrotal ultrasound to assess testicular volume, rule out intratesticular pathology, and confirm varicocele. Transrectal ultrasound (TRUS) if ejaculatory duct obstruction is suspected.
6. Genetic testing (when indicated): Karyotype (for azoospermia or severe oligozoospermia <5 million/mL), Y-chromosome microdeletion testing, CFTR mutation analysis (if CBAVD is found).
Treatment Options: What Can Actually Be Done
Treatment depends entirely on the cause — which is why proper diagnosis matters so much.
Varicocelectomy: Microsurgical repair for clinically significant varicocele with abnormal semen parameters. Improves parameters in 60–70% of men. Spontaneous pregnancy rate: 30–40%.
Hormonal therapy: Clomiphene citrate or hCG for men with hypogonadotropic hypogonadism or TRT-induced suppression. These agents stimulate the pituitary to produce LH and FSH, driving endogenous testosterone and sperm production.
Surgical sperm retrieval + ICSI: For obstructive or non-obstructive azoospermia. TESE or micro-TESE to obtain sperm, combined with IVF/ICSI for fertilization.
Vasectomy reversal (vasovasostomy): For post-vasectomy infertility. Success rates of 75–95% for sperm return and 30–75% for pregnancy, depending on the interval since vasectomy.
Lifestyle modification: For men with mild-moderate oligozoospermia and identifiable lifestyle factors. Weight loss, smoking cessation, heat avoidance, anabolic steroid cessation. Allow 3 months (one spermatogenesis cycle) to reassess.
Antioxidant supplementation: There is moderate-quality evidence that oral antioxidants (vitamin C, vitamin E, CoQ10, selenium, zinc, L-carnitine) may improve semen parameters and reduce sperm DNA fragmentation in subfertile men. The Males, Antioxidants, and Infertility (MOXI) trial was disappointing for live birth rates, but smaller studies and meta-analyses suggest benefit for semen parameters specifically. I consider antioxidants a low-risk adjunct — not a standalone treatment.
💡 In My Practice
The most common scenario I encounter is a couple who’ve been trying for 18–24 months, where the wife has undergone extensive gynecological evaluation — hormonal panels, HSG (hysterosalpingography), ultrasound, sometimes even diagnostic laparoscopy — before anyone ordered a semen analysis. When I finally see the husband, the result is often a clear abnormality: a grade 3 varicocele with oligoasthenozoospermia, or severe oligozoospermia from an undiagnosed hormonal issue. The delay cost the couple years of emotional distress and significant expense.
I’ve also seen a concerning rise in young men in their late 20s and 30s presenting with azoospermia or severe oligozoospermia caused by anabolic steroid use or unmonitored TRT from online clinics. These men were never warned that exogenous testosterone shuts down sperm production. Some recover after cessation and hCG/clomiphene therapy. Others face a long, uncertain road to recovery — and some may need micro-TESE and IVF to have biological children.
⚠️ When to See a Doctor — Urgently
- 12 months of unprotected intercourse without conception — this is the standard definition of infertility. If the female partner is over 35, seek evaluation after 6 months. Both partners should be investigated simultaneously
- A new testicular lump or swelling — while varicoceles are common, any new lump must be urgently investigated by ultrasound to exclude testicular cancer, which peaks in men aged 20–40
- Sudden testicular pain with swelling — testicular torsion is a surgical emergency requiring treatment within 6 hours. Epididymo-orchitis (infection) also requires prompt antibiotic treatment to prevent permanent damage to fertility
- History of undescended testis (cryptorchidism) in childhood — even if surgically corrected, cryptorchidism is a risk factor for both infertility and testicular cancer. Fertility should be assessed proactively if conception is planned
- Blood in the semen (hematospermia) for more than 4 weeks — while usually benign and self-limiting, persistent hematospermia warrants investigation to exclude infection, obstruction, or rarely, malignancy
Download Your Free Clinical Action Plan
Enter your email below to download Dr. Khalid’s complete Male Fertility Optimization Checklist as a free, printable PDF.
Frequently Asked Questions
Can a man with zero sperm count still have biological children?
Yes — in many cases. Azoospermia (zero sperm in the ejaculate) doesn’t necessarily mean zero sperm production. If the cause is obstructive (blocked vas deferens, ejaculatory duct obstruction), sperm can be retrieved surgically from the testis or epididymis (TESE, PESA) and used with IVF/ICSI. Even in non-obstructive azoospermia, where the testes are producing very little or no sperm, micro-TESE — a microsurgical procedure that searches for focal areas of spermatogenesis within the testis — has a sperm retrieval rate of 40–60% in experienced centers. The critical step is determining why there are no sperm. Check your exact numbers with our Semen Analysis Interpreter.
Does laptop heat actually damage sperm?
The evidence says yes. Spermatogenesis is exquisitely temperature-sensitive — it requires the scrotum to be 2–3°C below core body temperature, which is why the testes are external. Placing a laptop directly on the lap raises scrotal temperature by 1–2.8°C within 15–30 minutes, even with a lap pad underneath. A study published in Fertility and Sterility showed that regular laptop-on-lap use was associated with reduced sperm motility and increased DNA fragmentation. The practical advice: use a desk. If you must use a laptop on your lap, use a thick barrier and take frequent breaks.
How long after stopping testosterone injections will my sperm come back?
Recovery of spermatogenesis after stopping exogenous testosterone typically takes 6–12 months, though it can take longer — up to 24 months in some men. The speed of recovery depends on the dose used, the duration of use, and individual HPG axis resilience. hCG (human chorionic gonadotropin) and clomiphene citrate can accelerate recovery by stimulating LH and FSH production while your axis reboots. However, recovery is not guaranteed — a minority of men may have persistent oligozoospermia or azoospermia. Read our complete guide to Low Testosterone in Men Over 40.
Should men take supplements to improve sperm quality?
There is moderate-quality evidence that certain oral antioxidants — including vitamin C, vitamin E, CoQ10, selenium, zinc, and L-carnitine — may improve semen parameters, particularly in men with elevated sperm DNA fragmentation. However, the largest randomized controlled trial (the MOXI trial) found no significant improvement in live birth rates with antioxidant supplementation compared to placebo. My approach: antioxidants are a low-cost, low-risk adjunct that may help optimize sperm quality — but they are not a substitute for diagnosing and treating the underlying cause of infertility. Verify the evidence behind these supplements with our Supplement Matcher.
Is male infertility genetic?
It can be, particularly in severe cases. Genetic causes account for approximately 15–20% of severe male infertility. The most common genetic causes are: Klinefelter syndrome (47,XXY), present in about 1 in 600 men, causing primary testicular failure; Y-chromosome microdeletions, affecting regions (AZFa, AZFb, AZFc) critical for spermatogenesis; and CFTR gene mutations, which cause congenital bilateral absence of the vas deferens (CBAVD). Genetic testing is recommended for all men with azoospermia or severe oligozoospermia (sperm count below 5 million/mL).
📚 References
- Agarwal A, Mulgund A, Hamada A, Chyatte MR. A unique view on male infertility around the globe. Reprod Biol Endocrinol. 2015;13:37. [PubMed]
- World Health Organization. WHO laboratory manual for the examination and processing of human semen, 6th ed. Geneva: World Health Organization; 2021. [WHO Guidelines]
- Salonia A, Bettocchi C, Boeri L, et al. EAU Guidelines on Sexual and Reproductive Health. European Association of Urology. 2024. [EAU Guidelines]
- Baazeem A, Belzile E, Ciampi A, et al. Varicocele and male factor infertility treatment: a new meta-analysis and review of the role of varicocoelectomy in assisted reproductive technology. Eur Urol. 2011;60(4):796-808. [PubMed]
- Crosnoe LE, Grober E, Ohl D, Kim ED. Exogenous testosterone: a preventable cause of male infertility. Transl Androl Urol. 2013;2(2):106-113. [PubMed]
- Steiner B, et al. The impact of anabolic androgenic steroids on male fertility. Transl Androl Urol. 2021;10(3):1426-1435. [PubMed]
- Shefi S, Turek PJ. Definition and current evaluation of subfertile men. Int Braz J Urol. 2006;32(4):385-397. [PubMed]
- Corona G, Lee LS, Isidori AM, et al. Microdissection TESE versus conventional TESE in NOA. Hum Reprod Update. 2019;25(6):733-748. [PubMed]
- Garolla A, Torino M, Sartini B, et al. Seminal and molecular evidence that sauna exposure affects human spermatogenesis. Hum Reprod. 2013;28(4):877-885. [PubMed]
- Sharma R, Harlev A, Agarwal A, Esteves SC. Cigarette smoking and semen quality: a new meta-analysis. Eur Urol. 2016;70(4):635-645. [PubMed]
- Campbell JM, Lane M, Bowden J, et al. Semen quality and obesity. J Assist Reprod Genet. 2015;32(11):1579-1585. [PubMed]
- Hassan N, et al. The effect of age on semen parameters and male fertility. J Assist Reprod Genet. 2015;32(1):15-21. [PubMed]
- Steiner AZ, et al. Antioxidants in the treatment of male infertility (MOXI): a randomized controlled trial. Fertil Steril. 2020;113(3):552-560. [PubMed]
- Shefi S, et al. Laptop computers and scrotal temperature. Fertil Steril. 2011;95(2):647-651. [PubMed]

Dr. Muhammad Khalid
MBBS · FCPS (Urology) · MCPS (Gen. Surgery) · CHPE · CRSM · IMC #539472
Specialist urologist with 11+ years of clinical experience across tertiary teaching hospitals. Trained at Lady Reading Hospital and Khyber Teaching Hospital, Peshawar. Author of 5 peer-reviewed international publications in Cureus, WJSA, and AJBS. Procedural expertise: URS, PCNL, RIRS, TURP, TURBT, and major open urological surgery. Full profile →
This article is for educational purposes only and does not constitute medical advice. Always consult your physician or urologist for diagnosis and treatment decisions specific to your condition.
