
Prostate cancer screening is one of the most debated topics in modern medicine — and one of the most confusing for the men it actually affects. Should you get a PSA test at 45 or 50? What if your father had prostate cancer? What does it mean if your PSA is “slightly raised”? And why do some doctors recommend screening while others say it causes more harm than good?
The confusion exists because the answer genuinely isn’t simple. Prostate cancer screening can save lives — but it can also lead to unnecessary biopsies, overdiagnosis of cancers that would never have caused harm, and treatments with life-altering side effects for disease that didn’t need treating. The key is not whether to screen, but who to screen, when to start, and how to interpret the results intelligently.
As a urologist, I have this conversation with patients every week. This article gives you the same framework I use — an age-by-age guide that balances the genuine benefits of early detection against the real risks of over-investigation.
Want to read this later?
Enter your email below to download Dr. Khalid’s complete Prostate Health Guide as a free, printable PDF.
📋 Key Takeaways
- Prostate cancer is the most common non-skin cancer in men — 1 in 8 men will be diagnosed during their lifetime [1]
- PSA testing saves lives when used in the right population, but causes harm through overdiagnosis when applied indiscriminately
- Screening should be a shared decision between you and your doctor — not a routine test done without discussion
- High-risk men (family history, Black ethnicity) should discuss screening from age 40–45. Average-risk men from age 50
- A single PSA number is far less useful than PSA trends over time (velocity). A rising PSA matters more than an isolated elevated value
- MRI before biopsy reduces unnecessary biopsies by 28% and detects clinically significant cancers more accurately than blind biopsy [2]
- Not all prostate cancers need treatment — low-grade cancers can often be safely monitored with active surveillance
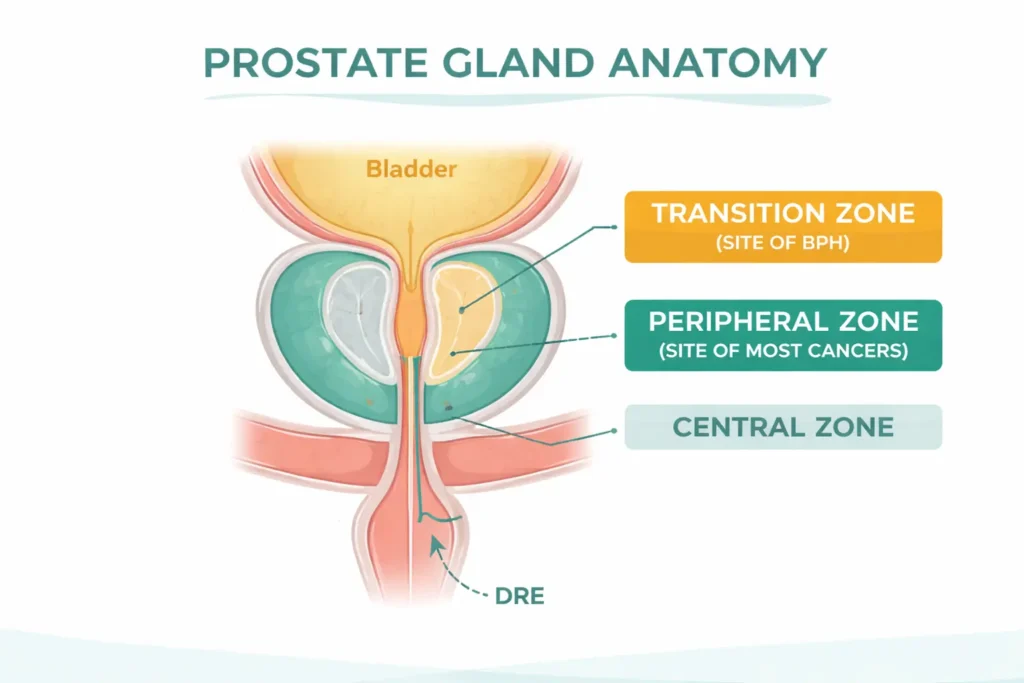
What Is Prostate Cancer Screening?
Prostate cancer screening consists of two components, though one has become far more central than the other:
PSA blood test (Prostate-Specific Antigen)
PSA is a protein produced by the prostate gland. It’s measured with a simple blood test. In healthy men, small amounts leak into the bloodstream. When the prostate is enlarged, inflamed, or cancerous, more PSA enters the blood, raising the level.
The critical thing to understand about PSA: it is prostate-specific, not cancer-specific. PSA can be elevated by benign prostatic hyperplasia (BPH), prostatitis, urinary tract infection, vigorous exercise, recent ejaculation, and even cycling — not just cancer. This is why an elevated PSA does not equal a cancer diagnosis. It’s a flag that says “investigate further,” not a verdict [3].
Digital rectal examination (DRE)
The doctor inserts a gloved, lubricated finger into the rectum to feel the back surface of the prostate. A normal prostate feels smooth, rubbery, and symmetrical. A concerning prostate may have hard nodules, asymmetry, or a fixed irregular surface.
DRE has limitations — it can only assess the posterior part of the prostate (cancers in the anterior zone are missed), and it detects mainly palpable disease. However, approximately 15–25% of prostate cancers are detected by DRE in men with a normal PSA [4]. Most urological guidelines therefore recommend DRE alongside PSA, not PSA alone.
The Age-by-Age Screening Guide
No major guideline recommends the same approach for every man at every age. Screening should be tailored to individual risk. Here’s how I approach it, aligned with the European Association of Urology (EAU) and American Urological Association (AUA) guidelines [5][6]:
Ages 40–44: High-risk men only
Who should consider screening at this age:
- Men with a first-degree relative (father, brother) diagnosed with prostate cancer, especially if diagnosed before age 65
- Men with two or more first-degree relatives with prostate cancer at any age
- Men of African or Caribbean descent — who have approximately double the risk of prostate cancer compared to white men, and are more likely to be diagnosed with aggressive disease at younger ages [7]
- Known BRCA2 mutation carriers — significantly elevated prostate cancer risk, especially aggressive forms
What to do: A baseline PSA test is reasonable. If PSA is below 1.0 ng/mL at age 40–44, the risk of developing clinically significant prostate cancer in the next 10–15 years is very low — repeat testing can be deferred to age 50. If PSA is above 1.0 ng/mL, consider testing every 2 years going forward [5].
This baseline value is surprisingly powerful. The EAU guidelines specifically note that a single PSA measurement at age 40–45 can stratify long-term prostate cancer risk more effectively than many years of later testing.
Ages 45–49: Expanding the net
Who should consider screening:
- All men in the high-risk categories listed above (if not started earlier)
- Men who are concerned about prostate cancer and want a baseline assessment after an informed discussion about the pros and cons
What to do: Offer a baseline PSA with DRE. If PSA is below 1.5 ng/mL, the risk is low — retest every 2–4 years. If PSA is 1.5–3.0 ng/mL, consider annual testing and closer monitoring. If above 3.0 ng/mL, further investigation is warranted.
Ages 50–69: The core screening window
This is where the strongest evidence for screening benefit exists. The European Randomized Study of Screening for Prostate Cancer (ERSPC) — the largest prostate cancer screening trial ever conducted, involving 182,000 men — demonstrated a 20% relative reduction in prostate cancer mortality with PSA-based screening in men aged 55–69, with up to 27% reduction at 16 years of follow-up [8].
What to do:
- Offer PSA testing with DRE every 2 years if PSA is below 2.0 ng/mL
- Offer annual testing if PSA is 2.0–3.0 ng/mL
- Investigate further if PSA is above 3.0 ng/mL or rising significantly
- This must be a shared decision. Every man in this age group should be offered the opportunity to discuss screening — but no man should be tested without understanding what an abnormal result means and what the next steps would involve
Ages 70–75: Individualised approach
Screening in this age group becomes more nuanced. The benefit of detecting prostate cancer needs to be weighed against life expectancy and competing health risks.
- Men in excellent health with a life expectancy of 10+ years: continued screening is reasonable, especially if PSA has been rising or was elevated in the 50–69 window
- Men with significant comorbidities (heart disease, diabetes, COPD) whose life expectancy is less than 10 years: screening is unlikely to benefit them and may cause harm through investigation and treatment of cancers that would never have caused symptoms
Ages 75+: Generally stop routine screening
Most guidelines recommend stopping routine PSA screening after age 75 (AUA) or when life expectancy is below 10 years (EAU). Prostate cancer is typically slow-growing, and the time from diagnosis to cancer-specific death is usually 10–15+ years for localised disease. Detecting cancer in a 78-year-old man with multiple comorbidities is more likely to cause anxiety and treatment-related morbidity than to save his life.
Exception: Men with previously elevated or rising PSA values should continue monitoring. Any man with new urinary symptoms should still be evaluated — this is symptom-driven investigation, not screening.
➡️ Related Read: Enlarged Prostate (BPH) — When Medication Fails and Surgery Becomes the Answer
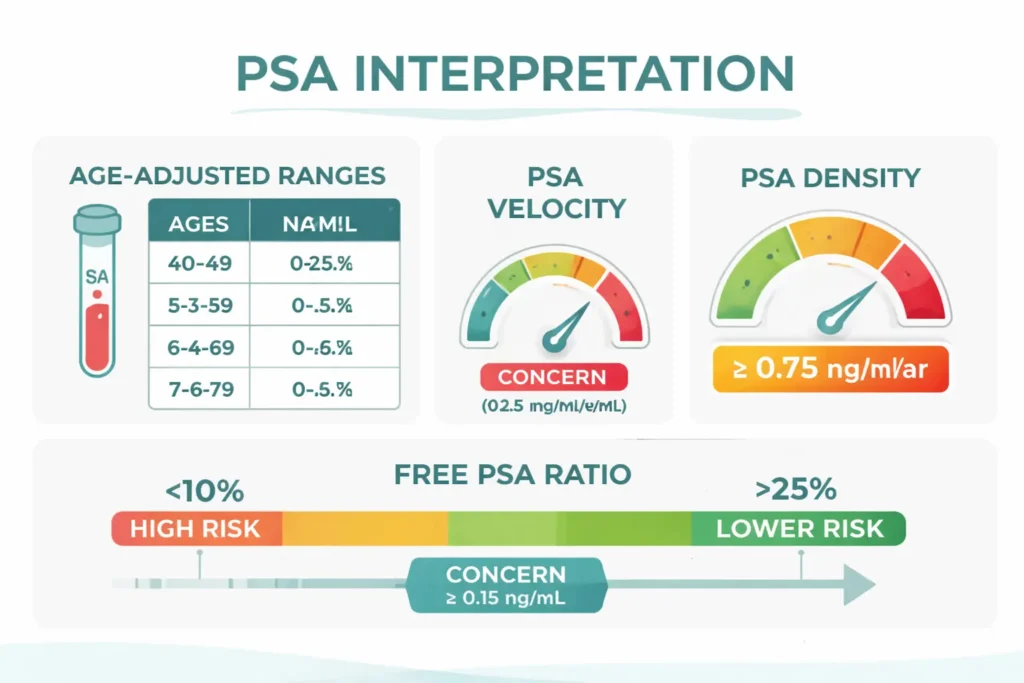
Understanding Your PSA Result: It’s Not Just a Number
The traditional “normal” cutoff for PSA was 4.0 ng/mL — anything above that triggered a biopsy. This binary approach has largely been abandoned because it’s too simplistic. Many cancers exist below 4.0, and many men above 4.0 don’t have cancer. Modern PSA interpretation uses multiple parameters:
Age-adjusted PSA ranges
PSA naturally rises with age as the prostate grows. What’s “normal” for a 45-year-old is different from what’s normal for a 70-year-old:
- 40–49 years: Normal range 0–2.5 ng/mL
- 50–59 years: Normal range 0–3.5 ng/mL
- 60–69 years: Normal range 0–4.5 ng/mL
- 70–79 years: Normal range 0–6.5 ng/mL [9]
A PSA of 3.8 in a 48-year-old is far more concerning than the same value in a 72-year-old with a 60-gram prostate.
PSA velocity (rate of change over time)
This is one of the most underused tools in clinical practice. A PSA that rises by more than 0.75 ng/mL per year is concerning, even if the absolute value is still within “normal” range. A man whose PSA goes from 1.2 to 2.8 in two years (velocity of 0.8/year) should be investigated, even though both numbers are technically “normal” [10].
Serial PSA measurements are more valuable than a single test. Your PSA trend tells a far more accurate story than any isolated number.
Free-to-total PSA ratio
PSA exists in the blood in two forms: bound to proteins (complexed) and free (unbound). Prostate cancer tends to produce more complexed PSA, so a low free-to-total ratio suggests higher cancer risk.
- Free PSA above 25%: Lower cancer risk — more likely BPH
- Free PSA below 10%: Higher cancer risk — biopsy strongly recommended
- Free PSA 10–25%: Grey zone — clinical judgement needed, consider MRI
Free-to-total ratio is most useful in the PSA 4–10 range (the “grey zone”) where the probability of cancer is approximately 25% — meaning 75% of biopsies done based on total PSA alone in this range find no cancer [3].
PSA density
PSA divided by prostate volume (measured on ultrasound or MRI). A larger prostate naturally produces more PSA. PSA density above 0.15 ng/mL/cc is more concerning for cancer than a high PSA from a very large benign prostate. A man with a PSA of 8.0 and a 100-gram prostate (density 0.08) is far less likely to have cancer than a man with a PSA of 8.0 and a 30-gram prostate (density 0.27) [11].
➡️ Related Read: My PSA is 4.5 — Do I Have Cancer? Understanding Grey Zone Results
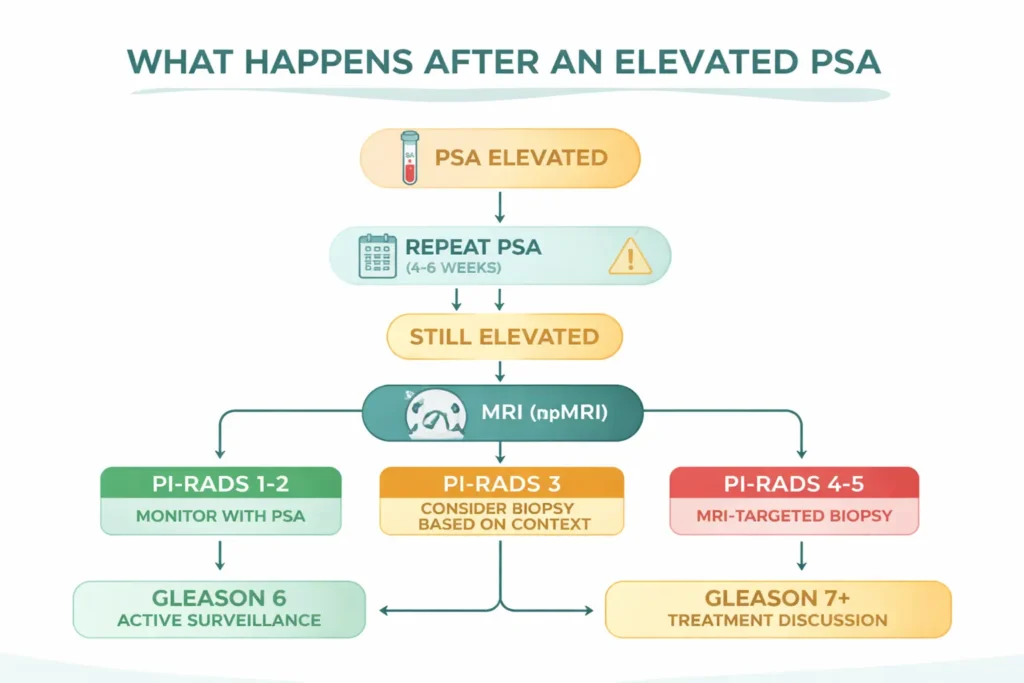
The MRI Revolution: Why Biopsy Isn’t Always the Next Step
Until recently, the pathway after an elevated PSA was straightforward: straight to biopsy. A TRUS (transrectal ultrasound) guided biopsy — 10–12 needle cores taken semi-blindly through the rectal wall — was the standard. This approach had two major problems: it missed cancers in areas the needles didn’t sample (particularly anterior cancers), and it detected many low-grade cancers that would never cause harm but then triggered treatment cascades.
Multiparametric MRI (mpMRI) has changed this paradigm fundamentally.
The landmark PROMIS trial (2017) and PRECISION trial (2018) demonstrated that performing an MRI before biopsy:
- Reduces unnecessary biopsies by 28% — if the MRI shows no suspicious lesion, biopsy can often be deferred with monitoring [2]
- Detects clinically significant cancer 18% more accurately than blind TRUS biopsy
- Reduces detection of clinically insignificant (harmless) cancers by 5% — fewer men diagnosed with cancers they don’t need to know about
The MRI uses a scoring system called PI-RADS (Prostate Imaging Reporting and Data System), scored 1–5:
- PI-RADS 1–2: Very low/low probability of clinically significant cancer. Biopsy generally not needed. Monitor with PSA
- PI-RADS 3: Equivocal — intermediate probability. Biopsy may be considered based on clinical context (PSA density, risk factors)
- PI-RADS 4–5: High/very high probability. MRI-targeted biopsy strongly recommended
When biopsy IS indicated after MRI, modern practice favours MRI-targeted biopsy (fusion biopsy) — the MRI images are overlaid onto the ultrasound during biopsy, allowing the urologist to target specific suspicious areas rather than sampling blindly. The transperineal approach (through the skin between the scrotum and anus, rather than through the rectum) is increasingly preferred because it virtually eliminates the risk of sepsis from rectal bacteria — a complication that occurs in 1–4% of TRUS biopsies [12].
The Overdiagnosis Problem: The Elephant in the Room
This is where prostate cancer screening becomes genuinely complicated, and where an honest discussion is owed rather than a simple recommendation.
The problem: Autopsy studies show that approximately 50% of men over 50 harbour microscopic prostate cancer [13]. The vast majority of these cancers are low-grade (Gleason 6 / Grade Group 1), slow-growing, and would never cause symptoms or death during the man’s natural lifetime. They would die with prostate cancer, not from it.
PSA screening detects some of these cancers. Once detected, the psychological burden of a cancer diagnosis — even a “low-risk” one — is significant. Many men opt for radical treatment (surgery or radiotherapy) for cancers that would have remained harmless. That treatment carries real side effects: erectile dysfunction (30–70% after radical prostatectomy), urinary incontinence (5–20%), and bowel problems with radiotherapy [14].
The ERSPC trial estimated that for every prostate cancer death prevented by screening, approximately 3 men were overdiagnosed — diagnosed with and treated for cancers that would never have harmed them [8]. This number has improved with the adoption of MRI-first pathways and active surveillance, but overdiagnosis remains a real concern.
The counterpoint: Prostate cancer is still the second leading cause of cancer death in men in many Western countries. The men who die from it are predominantly those diagnosed late, with high-grade or metastatic disease. Early detection of clinically significant cancer — Gleason 7 or higher (Grade Group 2+) — genuinely saves lives by allowing curative treatment before the disease spreads.
The balance: The goal of modern screening isn’t to find every cancer. It’s to find the dangerous ones early while avoiding detection of the harmless ones. The MRI-first approach has moved us significantly in this direction, but it’s not perfect. This is exactly why screening must be a shared, informed decision — not a reflexive box-ticking exercise.
Active Surveillance: When “Doing Nothing” Is the Right Treatment
One of the most important advances in prostate cancer management is the widespread adoption of active surveillance for low-risk disease. This is not “doing nothing” — it’s a structured monitoring programme designed to avoid treating cancers that don’t need treatment while catching any that progress before they become dangerous.
Who qualifies for active surveillance:
- Gleason 6 (Grade Group 1) cancer — the lowest grade
- PSA below 10 ng/mL
- Low volume of cancer on biopsy (typically fewer than 3 cores positive, with less than 50% of any core involved)
- Clinical stage T1c–T2a (non-palpable or small palpable nodule)
What active surveillance involves:
- PSA testing every 3–6 months
- DRE every 6–12 months
- Repeat MRI at 12 months, then periodically
- Repeat biopsy (confirmatory) at 12–18 months, then as indicated by PSA trends or MRI changes
- Intervention (surgery or radiotherapy) triggered only if the cancer shows signs of upgrading (Gleason score increase) or progression
Long-term data now supports this approach. The ProtecT trial (10-year follow-up) found that prostate cancer-specific mortality was extremely low (below 1%) regardless of whether men were treated immediately or monitored, though men on active surveillance had a slightly higher rate of developing metastatic disease [15]. This reinforces the importance of strict monitoring protocols and willingness to intervene if the disease progresses.
Active surveillance spares approximately 50–60% of eligible men from unnecessary treatment over 10 years — treatment that would have impaired their erectile function, continence, or both, for a cancer that was never going to harm them [15].
💡 In My Practice
The conversation I have most frequently about screening is with men in their early 50s who’ve been told by someone — a friend, a news article, a GP — that “PSA tests are unreliable” or “cause more harm than good.” While the concerns about overdiagnosis are legitimate, I think dismissing PSA testing entirely is dangerous. I’ve seen men present in their 60s with metastatic prostate cancer who were told years earlier that screening wasn’t worthwhile. That’s a man who might have been cured with early detection.
My approach is nuanced: I explain the benefits and limitations clearly, I offer the test, and I let the patient decide. If they choose screening, I use PSA velocity over time rather than reacting to a single number. And if PSA is elevated, I almost always recommend an MRI before biopsy — this single change has dramatically reduced the number of unnecessary biopsies in my practice while catching the cancers that actually matter.
The men I worry most about are those with strong family histories who dismiss their own risk. If your father or brother had prostate cancer, your risk is 2–3 times the average — and starting screening at 40–45 is genuinely important for this group. A baseline PSA at 45 is one of the cheapest and most effective risk stratification tools in medicine.
⚠️ When to See a Doctor — Urgently
Request an urgent referral or appointment if:
- Your PSA is above the age-adjusted normal range — especially if confirmed on a repeat test taken 4–6 weeks later (to exclude transient causes like UTI or prostatitis)
- Your PSA has risen significantly over serial measurements (velocity >0.75 ng/mL/year)
- Your GP found an abnormality on DRE — a hard nodule, asymmetry, or fixed prostate
- You have a strong family history (first-degree relative with prostate cancer) and haven’t had baseline screening
- New urinary symptoms — difficulty urinating, visible blood in urine (haematuria), or bone pain — which could indicate locally advanced or metastatic disease
- Diagnosed with low-risk prostate cancer and want to discuss whether active surveillance is appropriate for your specific pathology
Frequently Asked Questions
Does a high PSA mean I have prostate cancer?
No. The majority of men with an elevated PSA do NOT have prostate cancer. In the PSA 4-10 range (grey zone), only about 25% of biopsies reveal cancer. BPH, prostatitis, and other benign conditions are far more common causes of PSA elevation. An elevated PSA means investigate further, not you have cancer.
Can I have prostate cancer with a normal PSA?
Yes, though it is less common. Approximately 15% of men with PSA below 4.0 ng/mL harbour prostate cancer on biopsy, though the vast majority of these are low-grade, clinically insignificant tumours. This is why DRE is still recommended alongside PSA — it occasionally detects palpable cancers that PSA alone would miss.
How do I prepare for a PSA test?
Avoid ejaculation for 48 hours before the test. Avoid vigorous exercise or cycling for 48 hours. Do not have the test during an active urinary infection. If you have recently had a DRE, catheterisation, or prostate biopsy, wait 2-6 weeks as these can temporarily raise PSA.
My father had prostate cancer — when should I start prostate cancer screening?
Discuss baseline PSA testing with your doctor at age 40-45. Having a first-degree relative with prostate cancer approximately doubles your risk. If your father was diagnosed before age 65, or if you have two or more affected relatives, your risk is even higher and earlier screening is strongly recommended.
If I am diagnosed with low-grade prostate cancer, will I definitely need surgery?
Not necessarily. Low-grade prostate cancer (Gleason 6, Grade Group 1) is increasingly managed with active surveillance rather than immediate treatment. Studies show that 50-60% of men on active surveillance avoid treatment entirely over 10 years, with excellent cancer-specific survival. The decision depends on your specific pathology, PSA dynamics, MRI findings, and personal preferences.
📚 References
- Rawla P. Epidemiology of prostate cancer. World J Oncol. 2019;10(2):63–89.
- Kasivisvanathan V, et al. MRI-targeted or standard biopsy for prostate-cancer diagnosis (PRECISION trial). N Engl J Med. 2018;378(19):1767–1777.
- Catalona WJ, et al. Measurement of prostate-specific antigen in serum as a screening test for prostate cancer. N Engl J Med. 1991;324(17):1156–1161.
- Gosselaar C, et al. The value of digital rectal examination for screening for prostate cancer in a study with four-year follow-up. Eur Urol. 2009;55(4):804–812.
- EAU Guidelines on Prostate Cancer. European Association of Urology. 2024.
- Carter HB, et al. Early detection of prostate cancer: AUA Guideline. J Urol. 2013;190(2):419–426.
- Rebbeck TR, et al. Global patterns of prostate cancer incidence, aggressiveness, and mortality in men of African descent. Prostate Cancer. 2013;2013:560857.
- Schröder FH, et al. Screening and prostate cancer mortality: results of the ERSPC at 13 years of follow-up. Lancet. 2014;384(9959):2027–2035.
- Oesterling JE, et al. Serum prostate-specific antigen in a community-based population of healthy men: establishment of age-specific reference ranges. JAMA. 1993;270(7):860–864.
- Carter HB, et al. Longitudinal evaluation of prostate-specific antigen levels in men with and without prostate disease. JAMA. 1992;267(16):2215–2220.
- Benson MC, et al. Prostate specific antigen density: a means of distinguishing benign prostatic hypertrophy and prostate cancer. J Urol. 1992;147(3 Pt 2):815–816.
- Grummet JP, et al. Transperineal vs transrectal biopsy of the prostate: a review. Eur Urol Focus. 2020;6(4):677–680.
- Jahn JL, et al. The high prevalence of undiagnosed prostate cancer at autopsy. Int J Cancer. 2015;137(12):2795–2802.
- Wilt TJ, et al. Radical prostatectomy versus observation for localised prostate cancer (PIVOT trial). N Engl J Med. 2012;367(3):203–213.
- Hamdy FC, et al. 10-year outcomes after monitoring, surgery, or radiotherapy for localised prostate cancer (ProtecT trial). N Engl J Med. 2016;375(15):1415–1424.
- Thompson IM, et al. Prevalence of prostate cancer among men with a prostate-specific antigen level ≤4.0 ng per milliliter. N Engl J Med. 2004;350(22):2239–2246.

Dr. Muhammad Khalid
MBBS · FCPS (Urology) · MCPS (Gen. Surgery) · CHPE · CRSM · IMC #539472
Specialist urologist with 11+ years of clinical experience across tertiary teaching hospitals. Trained at Lady Reading Hospital and Khyber Teaching Hospital, Peshawar. Author of 5 peer-reviewed international publications in Cureus, WJSA, and AJBS. Procedural expertise: URS, PCNL, RIRS, TURP, TURBT, and major open urological surgery. Full profile →
This article is for educational purposes only and does not constitute medical advice. Always consult your physician or urologist for diagnosis and treatment decisions specific to your condition.
